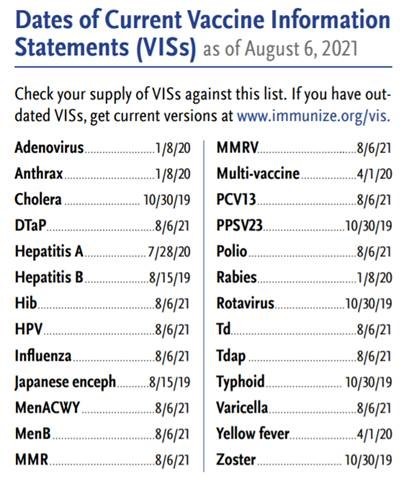
Vaccine Information Statements: What You Need to Know : Centers for Disease Control and Prevention : Free Download, Borrow, and Streaming : Internet Archive
Oregon Health Authority : Vaccine Information Statements : Vaccines and Immunization : State of Oregon
Improving vaccine-related health literacy in parents: comparison on the readability of CDC Vaccine Information Statements (VIS) and Health and Human Services (HHS) patient-facing vaccine literature - Beverley C. Millar, John E. Moore,